The FDA has sued a dietary supplement manufacturer, ATF Fitness Products and Manufacturing ATF Dedicated Excellence (MADE), for non-compliance with the Federal Food, Drug, and Cosmetic Act.
The company has been charged with, by the US Department of Justice, adulteration and mislabeling of its products, besides violation of current Good Manufacturing Practice (cGMP) regulations.
ATF buys dietary supplements including vitamins and minerals from MADE which markets them across the US under the brands Sci-Fit, Nature's Science and For Store Only.
Source: http://otc.pharmaceutical-business-review.com/news/fda-sues-dietary-supplement-maker-for-violating-laws-281111
News, musings and commentary on dietary supplements & pharmaceutical law issues, technology, and litigation. Lawyers for consumers and injured people.(No advice on this blog, though) mark(at)markzamora.com
Tuesday, November 29, 2011
Monday, November 28, 2011
Nevada Supreme Court
The case is Klasch adv. Walgreens, and the opinion may be found here.
In that case, a consumer had a prescription filled at a Walgreen's pharmacy. The consumer had completed a patient profile that indicated known allergies. When a prescription was to be filled, a Walgreens employee called the consumer (Klasch) and conveyed that her prescription had been flagged because of her sulfa allergy. In response, Klasch reportedly indicated that she had taken Bactrim in the past and that she had not experienced any adverse reaction to it. Satisfied with this clarification, a pharmacist then manually overrode the computer system’s flag, and the prescription was released. The consumer took the drug, and developed a horrible condition called SJS. She ultimately died from the complications of that condition, which the survivors in a lawsuit claim were caused by the prescription filled by Walgreens.
The complaint alleged that Walgreens’ pharmacist breached her duty of care by failing to adequately warn Klasch of the prescribed medication’s risks in light of her allergy to it or, alternatively, by failing to call her doctor to clarify whether he really meant to prescribe a medication to which she was allergic.
The Court held that : "The learned-intermediary doctrine does not foreclose a pharmacist’s potential for liability when the pharmacist has knowledge of a customer-specific risk."
The Court denied a Motion for Summary Judgment, in its ruling. It's worth reading.
Wednesday, November 23, 2011
Recent State Court decision detailing Post -Mensing Rulings (Reglan)
From a PA State Court, this decision does a good job noting recent post Mensing decisions:
Moss order11.18.11 order denying mensing mtd
View more documents from mzamoralaw.
Merck Agrees to Pay US $950 M to Settle Vioxx Claims
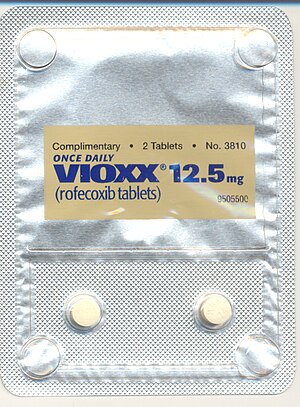 Image via WikipediaMerck will pay $950 million and a unit of the company will plead guilty to a criminal misdemeanor charge to resolve a U.S. probe of its illegal marketing of the painkiller Vioxx.
Image via WikipediaMerck will pay $950 million and a unit of the company will plead guilty to a criminal misdemeanor charge to resolve a U.S. probe of its illegal marketing of the painkiller Vioxx.The criminal plea is tied to Merck’s off-label promotion of rofecoxib for rheumatoid arthritis (RA) from 1999 until 2002, since the drug did not have an indication for RA at that time. The civil settlement is tied to a broader range of allegedly illegal conduct by Merck, including statements about the cardiovascular safety of rofecoxib made by Merck representatives. The company will also enter into an expansive corporate integrity agreement with the government.
From a press release:
Under civil settlement agreements signed with the United States and individually with 43 states and the District of Columbia, Merck will pay approximately two-thirds of the reserved charge to resolve civil allegations related to Vioxx. As a result, the United States and the participating states have released Merck from civil liability related to the governments' allegations regarding the sale and marketing of Vioxx in the United States. Previously disclosed litigation with seven states remains outstanding.
The civil settlement does not constitute any admission by Merck of any liability or wrongdoing.
"We believe that Merck acted responsibly and in good faith in connection with the conduct at issue in these civil settlement agreements, including activities concerning the safety profile of Vioxx," said Bruce N. Kuhlik, executive vice president and general counsel of Merck.
Separately, the company agreed to plead guilty to a misdemeanor under the Federal Food, Drug, and Cosmetic Act arising out of the marketing of Vioxx by company representatives to physicians in the United States for the treatment of rheumatoid arthritis before the FDA's approval of that indication in April 2002. The company will pay a fine of approximately one-third of the reserved amount to the federal government as part of the plea agreement.
Tuesday, November 22, 2011
Georgia November 21, 2011 Yaz News
 Image via WikipediaAs the litigation moves along in the cases involving the Yaz birth control products, there is this news from recent discovery responses:
Image via WikipediaAs the litigation moves along in the cases involving the Yaz birth control products, there is this news from recent discovery responses:Bayer may have considering marketing the birth- control pills for unapproved uses according to company e-mails.
Bayer reps discussed promoting the contraceptive known as Yaz, a spinoff of Yasmin, for treatment of all types of premenstrual syndrome. Salespeople for Bayer unit Berlex Laboratories Inc., acquired in the 2006 purchase of Schering AG, received an e-mail that year from a company official citing a Woman’s Day magazine article about Yaz.
From the email: “This article is a nice way of using YAZ for PMS treatment instead of just focusing on the specific” class of women battling premenstrual dysphoric disorder, the most severe form of PMS, wrote Matt Sample, a Berlex sales consultant, according to a copy of the e-mail produced as evidence.
The FDA approved Yaz only for the most severe form of PMS. Yaz is approved to treat Treat premenstrual dysphoric disorder (PMDD).
Premenstrual dysphoric disorder—PMDD—is the most severe form of PMS. That is, the only actual difference between the two syndromes is the severity of their symptoms. Even that is not always clear, since there are severe cases of PMS that do not happen to meet the criteria for PMDD.
One characteristic that can distinguish PMDD from PMS is the intensity of the emotional or psychological symptoms. In PMS, physical symptoms or emotional symptoms can be the primary cause of concern. The related cardinal feature of premenstrual dysphoric disorder is the degree to which it interferes with work and relationships. Of course, severe (or even moderate) PMS can have a tremendous impact on a woman's enjoyment of life and her productivity at work and at home. Source
The FDA warned that women taking the pills were 74 percent more likely to suffer blood clots than women on other low-estrogen contraceptives. The FDA examined data on 835,826 women who took pills containing the hormone, including Bayer’s Yasmin line of birth-control pills, according to the FDA report.
http://www.bloomberg.com/news/2011-11-21/bayer-may-have-touted-birth-control-pills-for-unapproved-use-e-mails-show.html#
Monday, November 21, 2011
Army drops use of anti-malarial drug
From the source below. The news today is that the Army will stop using Mefloquine. Mefloquine is used to treat malaria (a serious infection that is spread by mosquitoes in certain parts of the world and can cause death) and to prevent malaria in travelers who visit areas where malaria is common. Mefloquine is in a class of medications called antimalarials. It works by killing the organisms that cause malaria.
From the article:
Mefloquine is a zombie drug. It’s dangerous, and it should have been killed off years ago,” said Dr. Remington Nevin, an epidemiologist and Army major who has published research that he said showed the drug can be potentially toxic to the brain. He believes the drop in prescriptions is a tacit acknowledgement of the drug’s serious problems.
Over the past three years, the Army slashed by almost 75 percent the amount of mefloquine it prescribes, even as it sent thousands more soldiers to malaria-prone Afghanistan.
The decrease in doses followed two orders from military and Pentagon leaders in 2009. One, from the Army’s surgeon general, ordered the branch to limit its use to specific circumstances. Other branches, however, continue to favor mefloquine.
Friday, November 18, 2011
November 17, 2011 Update: encore100® Toric and encore100® Sphere Limited Lot Recall Q&As
 Image via Wikipedia
Image via WikipediaFrom the Cooper Vision Site:
encore100® Toric and encore100® Sphere Limited Lot Recall Q&As
Q: What is the cause of the encore100 Toric and encore100 Sphere recall?
A: The recall was initiated on limited lots of encore100 Toric and encore100 Sphere because
CooperVision identified certain lots that did not meet our updated quality requirements due to the
level of a residue (silicone oil). The presence of the residue may cause hazy vision or discomfort,
severe eye pain or eye injuries requiring medical treatment. Not everyone experiences the same
symptoms.
Q: What happens if a contact lens wearer puts an impacted lens in their eye?
A: We have received complaint reports for the encore100 Toric lenses that began as hazy vision.
After the recall, some patient complaints were received that began as hazy vision but progressed to
severe eye pain or eye injuries requiring medical treatment. Not every patient has the same
symptoms. Health and safety of patients are our top priority so the recall was expanded to the limited
lots of encore100 Sphere lenses.
Q: What should I do if I personally experience these symptoms?
A: Remove the lenses from your eyes immediately if you are currently wearing them. Contact your
eye care practitioner to let them know you are experiencing symptoms. Follow your eye care
practitioner’s instructions.
Q: If I have symptoms how long will they last?
A: If the lens is removed promptly after the onset of symptoms it likely will clear up within 15 to 30
minutes. If you leave the lens in your eye for a prolonged period of time the symptoms may progress
to a more serious condition. Contact your eye care practitioner to let them know you are
experiencing symptoms.
Q: Has anyone had permanent damage to their eye because of these symptoms?
A: To date we have not received any reports of permanent damage to a patient’s eye after wearing
the affected lens.
Q: What should be done with product that I currently have at home?
A: A small percentage of encore100 Toric and encore100 Sphere lenses are affected by this
situation. Please check the lot number on the carton or blister label you have by using the search
box on the www.coopervision.com/international-recalll website to see if your product has been
impacted by the recall, and if so, discontinue wear and return the lenses to your eye care
practitioner. You may also call our Consumer Care hotline at 1-855-526-6737.
Q: Will I be able to re-order my encore100 Toric and encore100 Sphere lenses?
A: We expect that encore100 Toric will be available for ordering in the spring of 2012. We anticipate
minimal disruption to availability of encore100 Sphere and you may continue to order through your
practitioner.
http://coopervision.com/pv_obj_cache/pv_obj_id_B84970CE53ADAF9E19484A5FD2C6ABD949630000/filename/encore100_Toric_and_encore100_Sphere_Consumer_FAQs_v7.pdf
Keime Inc dba Barry's Vitamins Conducts a Nationwide Voluntary Recall of Virility Max Dietary Supplement
 Image via WikipediaNovember 10, 2011 - Keime Incorporated announced today that it is conducting a voluntary recall of one lot of the companies dietary supplement product sold under the following name: Virility Max. The company has been informed by representatives of the FDA that lab analysis by FDA for Lot 10090571, found the product contained sulfoaildenafil, an analog of sildenafil. Sildenafil is an active ingredient of an FDA approved drug for erectile dysfunction (ED), making Virility Max an unapproved drug.
Image via WikipediaNovember 10, 2011 - Keime Incorporated announced today that it is conducting a voluntary recall of one lot of the companies dietary supplement product sold under the following name: Virility Max. The company has been informed by representatives of the FDA that lab analysis by FDA for Lot 10090571, found the product contained sulfoaildenafil, an analog of sildenafil. Sildenafil is an active ingredient of an FDA approved drug for erectile dysfunction (ED), making Virility Max an unapproved drug. Virility Max is used for sexual enhancement. It is distributed in 10 count, white plastic bottles to retail customers in the South Florida area.
No illnesses have been reported to the company to date in connection with this product.
Customers who have this product in their possession should stop using it immediately and contact their physician if they have experienced any problems that may be related to taking this produc
Breast Cancer: FDA Revokes Avastin Approval
 Image via WikipediaFrom ABC:
Image via WikipediaFrom ABC:The U.S. Food and Drug Administration today announced it will revoke the approval of Avastin for breast cancer, citing a lack of evidence that the benefits outweigh the risks.
Avastin was approved for metastatic breast cancer in February 2008 under the agency's accelerated approval program, which offers patients early access to promising drugs while confirmatory clinical trials are carried out.
"Unfortunately the additional studies failed to confirm Avastin's initial promise," FDA Commissioner Dr. Margaret Hamburg said today.
The additional studies, carried out by Avastin maker Genentech, found only a small effect on tumor growth and no evidence that patients lived longer than they would taking standard chemotherapy. Because the drug can cause severe high blood pressure, bleeding, heart failure and perforations in the nose, stomach and intestines, an FDA advisory panel in June recommended revoking its approval.
Wednesday, November 16, 2011
Cooper Expands Recall Of Avaira Lenses
 Image via Wikipedia
Image via WikipediaThe Cooper Companies contact lenses unit CooperVision expanded a recall of some of its Avaira brand lenses. Under the expanded recall, CooperVision is now including limited lots of Avaira Sphere contact lenses, in addition to limited units of Avaira Toric lenses.
Cooper, along with the FDA, found certain lots of Avaira Sphere lenses did not meet new quality requirements linked to silicone oil residue levels, and plans to replace them with available inventory.
Kimberly-Clark Recalls Kotex Tampons
Kimberly Clark has recalled about 1,400 cases of Kotex tampons out of fear they may be contaminated with bacteria.
The recall applies specifically to 18-count and 36-count boxes of Kotex Natural Balance Security Unscented Tampons Regular Absorbency with sku numbers 15063 and 15068. All other Natural Balance tampons with other SKU numbers are safe to use, the company said.
The recalled tampons were sold at Walmart, Fry's and Smith's stores in Texas, Iowa, Kansas, Missouri, Nebraska, New Mexico, Arizona and Utah.
Tuesday, November 15, 2011
Mizuho OSI Modular Table Systems Recall
From the FDA site:
Product: OSI Modular Table Systems (including Modular Bases, Jackson Spinal, Imaging, Lateral, & Orthopedic Tops)
Manufacturing From: January 1992 - June 2011
Distribution From: January 1992 - June 2011
The affected models and lot numbers can be found below:
| Model | Product Name |
|---|---|
| 5803/5803I | Advanced Control Modular Base, 120VAC 60Hz or 230VAC 50/60Hz |
| 5890 | Retractable “I” Base - Manual tilt & lock functions |
| 5891 | Non-Retractable “I” Base - Manual tilt & lock functions |
| 5892 | Advanced Control Retractable “I” Base - electric powered tilt & lock |
| 5855 | Orthopedic Trauma Table Top |
| 5895 | Maximum Access Lateral Top |
| 5927 | Radiolucent Imaging Top with Tempur-Med |
| 5840-726 | Imaging Top w/ Tempur-med pad (part of Model 5827 System) |
| 5840-831 | Spinal Surgery Table Top (part of Jackson Spinal Surgery Top System) |
| 5943 | Jackson Spinal Table |
| 5843AP | Jackson Spinal Surgery Top w/Advanced Control Pad System 100/120VAC 50/60Hz |
| 5943API | Jackson Spinal Surgery Top w/Advanced Control Pad System 230VAC 50/60Hz |
| 5996/5996I | Advanced Control Pad System Variable Speed |
| 6977 | AXIS Jackson System with Advanced Control Pad System 100/230 VAC 50/60Hz |
Use: For patient positioning during a diverse set of orthopedic trauma, thoracic, and spinal surgery procedures.
Recalling Firm:
Mizuho Orthopedic Systems Inc
30031 Ahem Avenue
Union City, CA 94587
Mizuho Orthopedic Systems Inc
30031 Ahem Avenue
Union City, CA 94587
Reason for Recall: There are reports of injury related incidents while using Mizuho OSI Modular Table Systems. The incorrect removal of the T-pins that support the bottom base, instead of the T-pins that support the top, may result in the lower table top and patient falling to the floor. Another potential concern is unexpected movement/tilting of the table which may result in unanticipated movement and/or patient falls during surgery. Both of the above may result in serious injury or death
Pine Nut Recall Sees Second Lawsuit
 Image via Wikipedia
Image via Wikipedia
The Food and Drug Administration (FDA) is warning consumers not to eat Turkish pine nuts distributed by Sunrise Commodities, based in Englewood Cliffs, New Jersey, after FDA tests confirmed the presence of Salmonella on the product.FDA is collaborating with the Centers for Disease Control and Prevention (CDC) and State public health and agriculture officials to investigate a multistate outbreak of Salmonella Enteritidis infections. To date, the CDC reports there are at least 42 illnesses associated with the outbreak in Maryland, New Jersey, New York, Pennsylvania and Virginia.As part of FDA’s investigation, the Agency collected and tested samples of Turkish pine nuts from a warehouse used by Sunrise Commodities and from a warehouse used by a customer of Sunrise Commodities. Additional testing by the FDA confirmed Salmonella Enteritidis, matching the outbreak strain, was found on FDA's positive samples of pine nuts. FDA's State partners also collected samples of pine nuts distributed by Sunrise Commodities; some of those samples tested positive for SalmonellaEnteritidis and matched the outbreak strain.
Sunrise Commodities has voluntarily recalled four lots of the implicated product, totaling more than 21,000 pounds of pine nuts. Each lot was packed in 22-pound boxes and included the markings:- Warehouse Lot 27963 with the identifying code “PO#: 50165”
- Warehouse Lot 29628 with the identifying code “PO#: 50558”
- Warehouse Lot 27713 with the identifying code “PO#: 49595”
- Warehouse Lot 27427 with the identifying code “PO#: 50032”
Sunrise Commodities distributed the Turkish pine nuts in bulk to various food vendors in Florida, New Jersey, New York and Canada. Sunrise Commodities issued a recall notification to its customers dated November 3, 2011, alerting them of the test results and of the epidemiologic investigation and asking them to notify their subsequent customers of the recall.
Already lawsuits have been filed:Kimberly Waterbury filed suit Tuesday in federal court on behalf of herself and her minor child against American Pistachio Commodity Corp., which does business as Sunrise Commodities of Englewood Cliffs, New JerseyThe suit claims that Sunrise Commodities was strictly liable for the contaminated product. The suit also claims the company was negligent for not using reasonable care in the manufacture, distribution and sale of the product, and for violating federal, state and/or local food safety regulations.
http://www.democratandchronicle.com/article/20111115/NEWS01/111115031
The Food and Drug Administration (FDA) is warning consumers not to eat Turkish pine nuts distributed by Sunrise Commodities, based in Englewood Cliffs, New Jersey, after FDA tests confirmed the presence of Salmonella on the product.
FDA is collaborating with the Centers for Disease Control and Prevention (CDC) and State public health and agriculture officials to investigate a multistate outbreak of Salmonella Enteritidis infections. To date, the CDC reports there are at least 42 illnesses associated with the outbreak in Maryland, New Jersey, New York, Pennsylvania and Virginia.
As part of FDA’s investigation, the Agency collected and tested samples of Turkish pine nuts from a warehouse used by Sunrise Commodities and from a warehouse used by a customer of Sunrise Commodities. Additional testing by the FDA confirmed Salmonella Enteritidis, matching the outbreak strain, was found on FDA's positive samples of pine nuts. FDA's State partners also collected samples of pine nuts distributed by Sunrise Commodities; some of those samples tested positive for SalmonellaEnteritidis and matched the outbreak strain.
Sunrise Commodities has voluntarily recalled four lots of the implicated product, totaling more than 21,000 pounds of pine nuts. Each lot was packed in 22-pound boxes and included the markings:
- Warehouse Lot 27963 with the identifying code “PO#: 50165”
- Warehouse Lot 29628 with the identifying code “PO#: 50558”
- Warehouse Lot 27713 with the identifying code “PO#: 49595”
- Warehouse Lot 27427 with the identifying code “PO#: 50032”
Sunrise Commodities distributed the Turkish pine nuts in bulk to various food vendors in Florida, New Jersey, New York and Canada. Sunrise Commodities issued a recall notification to its customers dated November 3, 2011, alerting them of the test results and of the epidemiologic investigation and asking them to notify their subsequent customers of the recall.
http://www.democratandchronicle.com/article/20111115/NEWS01/111115031
Study Suggests Multaq Doubled Deaths From Heart Disease
Multaq - a drug that is approved to treat patients whose hearts intermittently race with quick and inefficient contractions may have double the risk of serious injury for those who have some type of erratic heart rhythm, according to a recent study.
A Multaq trial of more than 3200 patients was recently halted, supposedly because of the dangers to trial participants.
The study was paid for by Paris-based Sanofi and published in the New England Journal of Medicine. There were 21 deaths from cardiovascular causes in the Multaq group, compared with 10 among those getting placebo.
From the NEJM:
BACKGROUND
Dronedarone restores sinus rhythm and reduces hospitalization or death in intermittent atrial fibrillation. It also lowers heart rate and blood pressure and has antiadrenergic and potential ventricular anti-arrhythmic effects. We hypothesized that dronedarone would reduce major vascular events in high-risk permanent atrial fibrillation.
METHODS
We assigned patients who were at least 65 years of age with at least a 6-month history of permanent atrial fibrillation and risk factors for major vascular events to receive dronedarone or placebo. The first coprimary outcome was stroke, myocardial infarction, systemic embolism, or death from cardiovascular causes. The second coprimary outcome was unplanned hospitalization for a cardiovascular cause or death.
RESULTS
After the enrollment of 3236 patients, the study was stopped for safety reasons. The first coprimary outcome occurred in 43 patients receiving dronedarone and 19 receiving placebo (hazard ratio, 2.29; 95% confidence interval [CI], 1.34 to 3.94; P=0.002). There were 21 deaths from cardiovascular causes in the dronedarone group and 10 in the placebo group (hazard ratio, 2.11; 95% CI, 1.00 to 4.49; P=0.046), including death from arrhythmia in 13 patients and 4 patients, respectively (hazard ratio, 3.26; 95% CI, 1.06 to 10.00; P=0.03). Stroke occurred in 23 patients in the dronedarone group and 10 in the placebo group (hazard ratio, 2.32; 95% CI, 1.11 to 4.88; P=0.02). Hospitalization for heart failure occurred in 43 patients in the dronedarone group and 24 in the placebo group (hazard ratio, 1.81; 95% CI, 1.10 to 2.99; P=0.02).
CONCLUSIONS
Dronedarone increased rates of heart failure, stroke, and death from cardiovascular causes in patients with permanent atrial fibrillation who were at risk for major vascular events. Our data show that this drug should not be used in such patients. (Funded by Sanofi-Aventis; PALLAS ClinicalTrials.gov number, NCT01151137.)
Friday, November 11, 2011
Under Armour Recalls Chin Straps for Football Helmets -- Laceration Hazard
 Image via Wikipedia
Image via WikipediaWASHINGTON, D.C. - The U.S. Consumer Product Safety Commission, in cooperation with the retailer named below, today announced a voluntary recall of the following consumer product. Consumers should stop using recalled product immediately unless otherwise instructed. It is illegal to resell or attempt to resell a recalled consumer product.
Name of Product: UA Defender Chin Straps
Units: About 541,000
Retailer: Under Armour, Inc., of Baltimore, Md.
Importer: JR286, Inc., of Redondo Beach, Calif.
Hazard: The metal snap that connects the chin strap to the helmet has sharp edges, posing a laceration hazard when the user’s metal snap comes into contact with another player.
Incidents/Injuries: Under Armour has received six reports of injuries that required stitches.
Description: This recall involves all UA Defender Chin Straps. They were sold in the following colors: white and black, red, midnight, navy and royal and have a hard nylon shell, a padded chin area and a plastic strap with “Under Armour” printed on it. The chin straps have metal clasps that tighten the straps and attach them to the helmet.
Sold at: Sporting goods stores and Under Armour outlet stores nationwide and at www.underarmour.com from January 2008 through September 2011 for about $20.
Manufactured in: China
Remedy: Consumers should immediately stop using the recalled chin straps and contact Under Armour for a free replacement chin strap.
Consumer Contact: For additional information, contact Under Armour
Veteran's Day Thank You
 Image by lukemontague via Flickr
Image by lukemontague via FlickrIt's hard to put into words sometimes how you feel, how to say thanks. Today is not one of those days. My father in law was a World War II bomber pilot, and I have family in the Navy as well as the Army. Our family is grateful to all who have served. From those in the greatest generation, to those now in harm's way anywhere our US Forces are ... thank you.
A friend of the family - in his 40's, two kids - was called to serve in Afghanistan, and he did two tours. You can't fathom the fear and anguish in a family or the military man until you see it firsthand.
You keep us free, you protect us, you are willing to die for us. My family and I am grateful and pray for your safety.
Wednesday, November 09, 2011
Public Citizen Calls on FDA to Require Black Box Warning on Label for the Antibiotic Tigecycline
From PC:
The FDA should place a black box warning on an antibiotic used to treat a wide range of infections because of a higher mortality rate associated with taking the drug than other widely used antibiotics, says Public Citizen.
The antibiotic, tigecycline, also known as Tygacil, was approved by the FDA in 2005 to treat complicated skin infections and intra-abdominal infections and in 2009 to treat community-acquired pneumonia.
In 2010, the FDA issued a safety alert to health care professionals informing them of an increased mortality rate associated with the use of intravenous tigecycline compared to that of other antibiotics. The agency said that alternatives to tigecycline should be considered in patients with serious infections. At the same time, the FDA required that the drug label be modified to include data about the increased mortality risk, but these warnings were inadequate, Public Citizen said in the petition. Critical safety information does not appear in a black box warning at the top of the label and fails to describe appropriate restrictions on the use of the drug.
“Given the approximately 30 percent higher rate of mortality in subjects receiving tigecycline, as well the significantly higher rates of failures to cure serious infections, in comparison to subjects treated with comparator antibiotics, the content and placement of the warnings in the current drug label are woefully insufficient,” said Dr. Michael Carome, deputy director of Public Citizen’s Health Research Group. “Because of the increased death rate and the likely inappropriate use of tigecycline as a first-line, rather than last-resort, antibiotic for serious infections without simultaneous administration of other antibiotics, it is inexcusable that there is currently no black box warning for tigecycline. Such a warning, accompanied by stronger warnings describing appropriate restrictions on its use in other parts of the label, clearly is warranted to more effectively advise physicians about the dangers of using tigecycline to treat serious infections and to better protect public health.”
Public Citizen calls on the FDA to add a black box warning to the antibiotic’s label indicating the increased risk of death and advising that it should be used only as a last-resort antibiotic and then only in combination with one or more bactericidal antibiotics (antibiotics that directly kill bacteria, rather than interfere with bacteria growth). Public Citizen also is petitioning the agency to require an FDA-approved medication guide to be distributed to patients, as well as a “Dear Doctor” letter to physicians, warning them of the dangers of the drug and the need for restricted use.
http://www.citizen.org/pressroom/pressroomredirect.cfm?ID=3440
The FDA should place a black box warning on an antibiotic used to treat a wide range of infections because of a higher mortality rate associated with taking the drug than other widely used antibiotics, says Public Citizen.
The antibiotic, tigecycline, also known as Tygacil, was approved by the FDA in 2005 to treat complicated skin infections and intra-abdominal infections and in 2009 to treat community-acquired pneumonia.
In 2010, the FDA issued a safety alert to health care professionals informing them of an increased mortality rate associated with the use of intravenous tigecycline compared to that of other antibiotics. The agency said that alternatives to tigecycline should be considered in patients with serious infections. At the same time, the FDA required that the drug label be modified to include data about the increased mortality risk, but these warnings were inadequate, Public Citizen said in the petition. Critical safety information does not appear in a black box warning at the top of the label and fails to describe appropriate restrictions on the use of the drug.
“Given the approximately 30 percent higher rate of mortality in subjects receiving tigecycline, as well the significantly higher rates of failures to cure serious infections, in comparison to subjects treated with comparator antibiotics, the content and placement of the warnings in the current drug label are woefully insufficient,” said Dr. Michael Carome, deputy director of Public Citizen’s Health Research Group. “Because of the increased death rate and the likely inappropriate use of tigecycline as a first-line, rather than last-resort, antibiotic for serious infections without simultaneous administration of other antibiotics, it is inexcusable that there is currently no black box warning for tigecycline. Such a warning, accompanied by stronger warnings describing appropriate restrictions on its use in other parts of the label, clearly is warranted to more effectively advise physicians about the dangers of using tigecycline to treat serious infections and to better protect public health.”
Public Citizen calls on the FDA to add a black box warning to the antibiotic’s label indicating the increased risk of death and advising that it should be used only as a last-resort antibiotic and then only in combination with one or more bactericidal antibiotics (antibiotics that directly kill bacteria, rather than interfere with bacteria growth). Public Citizen also is petitioning the agency to require an FDA-approved medication guide to be distributed to patients, as well as a “Dear Doctor” letter to physicians, warning them of the dangers of the drug and the need for restricted use.
http://www.citizen.org/pressroom/pressroomredirect.cfm?ID=3440
FDA: Warnings on Methylene Blue, Linezolid, and Serotonergics
The FDA has updated warning to avoid prescribing either methylene blue or linezolid (Zyvox, Pfizer) in combination with serotonergic agents because of the potential to cause serotonin syndrome.
Today, the FDA announced that the risk for this harmful interaction appears to be confined, in general, to 2 specific types of serotonergic drugs: selective serotonin reuptake inhibitors and serotonin-norepinephrine reuptake inhibitors.
Methylene blue is a dye used in diagnostic procedures and in the treatment of conditions ranging from cyanide poisoning to methemoglobinemia. Linezolid is an antibacterial agent. Both drugs inhibit the action of monoamine oxidase A, an enzyme that breaks down serotonin. This allows the levels of the neurotransmitter to rise.
When serotonin builds up to excessive levels in patients taking serotonergic agents, the result can be serotonin syndrome. Its signs and symptoms include confusion, hyperactivity, memory problems, and other mental changes; muscle twitching, excessive sweating, shivering, or shaking; diarrhea; trouble with coordination; and fever.
http://www.medscape.com/viewarticle/751978
Today, the FDA announced that the risk for this harmful interaction appears to be confined, in general, to 2 specific types of serotonergic drugs: selective serotonin reuptake inhibitors and serotonin-norepinephrine reuptake inhibitors.
Methylene blue is a dye used in diagnostic procedures and in the treatment of conditions ranging from cyanide poisoning to methemoglobinemia. Linezolid is an antibacterial agent. Both drugs inhibit the action of monoamine oxidase A, an enzyme that breaks down serotonin. This allows the levels of the neurotransmitter to rise.
When serotonin builds up to excessive levels in patients taking serotonergic agents, the result can be serotonin syndrome. Its signs and symptoms include confusion, hyperactivity, memory problems, and other mental changes; muscle twitching, excessive sweating, shivering, or shaking; diarrhea; trouble with coordination; and fever.
http://www.medscape.com/viewarticle/751978
Tuesday, November 08, 2011
Zyprexa: First Verdict is for Eli Lilly
Eli Lilly & Co. isn’t responsible for the death of college student who was taking the drugmaker’s Zyprexa antipsychotic medication, a jury ruled in the first case to go to trial over the drug.
Jurors in state court in Los Angeles deliberated about 11 hours over two days before clearing Lilly of responsibility for the 2007 death of Cody Tadai, a 20-year-old community college student who succumbed to diabetes-related illnesses while taking Zyprexa for mental illness. His family alleged the company hid the drug’s diabetes risks to protect sales.
Monday, November 07, 2011
FDA has approved Xarelto bloodthinner for atrial fibrillation
From the FDA:
The U.S. Food and Drug Administration today approved the anti-clotting drug Xarelto (rivaroxaban) to reduce the risk of stroke in people
The safety and efficacy of Xarelto were evaluated in a clinical trial with more than 14,000 patients comparing Xarelto with the anti-clotting drug warfarin. In the trial, Xarelto was similar to warfarin in its ability to prevent stroke.
The U.S. Food and Drug Administration today approved the anti-clotting drug Xarelto (rivaroxaban) to reduce the risk of stroke in people
who have abnormal heart rhythm (non-valvular atrial fibrillation).
Atrial fibrillation occurs in more than 2 million Americans and is one of the most common types of abnormal heart rhythm. In atrial fibrillation, the beating of the heart’s two upper heart chambers (atria) is irregular and poorly coordinated. This leads to blood pooling in these chambers, resulting in blood clots. Non-valvular atrial fibrillation refers to atrial fibrillation in patients who do not have significant problems in their heart valves.
“Atrial fibrillation can lead to the formation of blood clots, which can travel to the brain, blocking blood flow and causing a disabling stroke,” said Norman Stockbridge, M.D., Ph.D., director of the Division of Cardiovascular and Renal Products in the FDA’s Center for Drug Evaluation and Research. “This approval gives doctors and patients another treatment option for a condition that must be managed carefully.”
A stroke occurs if the flow of blood to a portion of the brain is blocked. If brain cells die or are damaged because of a stroke, symptoms occur in the parts of the body that these brain cells control. Stroke symptoms include sudden weakness; paralysis or numbness of the face, arms, or legs; trouble speaking or understanding speech; and trouble seeing.
The safety and efficacy of Xarelto were evaluated in a clinical trial with more than 14,000 patients comparing Xarelto with the anti-clotting drug warfarin. In the trial, Xarelto was similar to warfarin in its ability to prevent stroke.
For people taking the drug for atrial fibrillation, Xarelto should be taken one time a day with the evening meal so that it will be completely absorbed.
As with other anti-clotting drugs, Xarelto can cause bleeding that, rarely, can lead to death. Bleeding was the most common adverse event reported by patients treated with Xarelto in the major clinical trial for the prevention of stroke in non-valvular atrial fibrillation. In that trial, Xarelto’s risk of major bleeding was similar to that of warfarin; however, it caused less bleeding into the brain and more bleeding into the stomach and intestines.
Xarelto has a boxed warning to make clear that people using the drug should not discontinue it before talking with their health care professional. Discontinuing the drug can increase the risk of stroke.
Subscribe to:
Comments (Atom)










