From various sources, including the FDA website, Yahoo, and Reuters:
Parents and doctors were told this week not to give drugs that contain promethazine hydrochloride to children younger than 2 years of age. There are reprots of seven cases of death linked to use of the antihistamine.
The FDA's safety alert also noted that it also has received 22 reports of severe breathing problems associated with use of the allergy drug, all in children younger than two years of age.
The drug (also labelled Promethazine HCl) is sold by Wyeth. The warning covers all forms of the drug - Syrups, suppositories, tablets and injectable liquids.
According to the FDA, labels on all versions of the drug have been updated to reflect the new warnings.
For more information go here
News, musings and commentary on dietary supplements & pharmaceutical law issues, technology, and litigation. Lawyers for consumers and injured people.(No advice on this blog, though) mark(at)markzamora.com
Friday, April 28, 2006
Thursday, April 27, 2006
SC Trial Blog: An Award and Good PR
David Swanner recently won a Silver Internet Marketing Association award, for his blog - sctriallaw.com. He was the only "solo" to win an award. IMA wrote about David's site:
I bring you a stalwart effort from Myrtle Beach, South Carolina. I love SC—spend a week in HHI every year. From the clever biblical domain (IMA does have a real name from the Old Testament) to the incorporation with a related SC trial law blog, being a solo personal injury lawyer need not be dull…online. And he can attest to the site bringing in something called revenue. Think about that.
David’s site was also recently named one of the best small law firm Web sites in South Carolina by Lawyers Weekly.
Well done, Mr. Swanner. His blog is a must have on your blog reader.
I bring you a stalwart effort from Myrtle Beach, South Carolina. I love SC—spend a week in HHI every year. From the clever biblical domain (IMA does have a real name from the Old Testament) to the incorporation with a related SC trial law blog, being a solo personal injury lawyer need not be dull…online. And he can attest to the site bringing in something called revenue. Think about that.
David’s site was also recently named one of the best small law firm Web sites in South Carolina by Lawyers Weekly.
Well done, Mr. Swanner. His blog is a must have on your blog reader.
Is the Merck Vioxx Liability "Manageable?"
From Forbes online, a UBS Investment Research Analyst predicts that for Merck, the estimated total liability as to Vioxx would be approximately $10 billion, which Patel said was "manageable" for Merck given its large cash reserves.
He also wrote that one "concerning" aspect of the verdict is that Merck "failed to provide adequate warning to prescribing physicians of an association between Vioxx and an increased risk of serious cardiovascular events." Go here.
My comment: For those in the USA that think that big bad trial lawyers are pushing this wagon, they are wrong. Merck has a worldwide problem with Vioxx and the alleged harm it has done. Were is the discussion concerning this global problem?
In December of 2005, a Beijing-based law firm said it would a collective lawsuit against US pharmaceutical company Merck & Co Inc next year, and is looking for Chinese users of the pain relief drug Vioxx who have suffered from side effects.
Also last year, according to Ynetnews.com, Israeli citizens planned to sue Merck in the United States.
Canadian citizens were set to file a class action against Merck, and similar lawsuits by residents of France, Italy, UK, and Australia had been filed or would be filed. .
Attorneys General are either filing suit against Merck or will file suit over Vioxx. This past July, Texas Attorney General Greg Abbott sued Merck on behalf of Texas citizens, saynig that the company falsely touted the safety of the drug, knowing it caused a higher risk of heart attack and cardiovascular problems.
He also wrote that one "concerning" aspect of the verdict is that Merck "failed to provide adequate warning to prescribing physicians of an association between Vioxx and an increased risk of serious cardiovascular events." Go here.
My comment: For those in the USA that think that big bad trial lawyers are pushing this wagon, they are wrong. Merck has a worldwide problem with Vioxx and the alleged harm it has done. Were is the discussion concerning this global problem?
In December of 2005, a Beijing-based law firm said it would a collective lawsuit against US pharmaceutical company Merck & Co Inc next year, and is looking for Chinese users of the pain relief drug Vioxx who have suffered from side effects.
Also last year, according to Ynetnews.com, Israeli citizens planned to sue Merck in the United States.
Canadian citizens were set to file a class action against Merck, and similar lawsuits by residents of France, Italy, UK, and Australia had been filed or would be filed. .
Attorneys General are either filing suit against Merck or will file suit over Vioxx. This past July, Texas Attorney General Greg Abbott sued Merck on behalf of Texas citizens, saynig that the company falsely touted the safety of the drug, knowing it caused a higher risk of heart attack and cardiovascular problems.
Prempro (Hormone Therapy): 1st Trial - Summer 2006
Wyeth is up to bat as the next Big Pharma company having to defend one of its drugs:
In June, the first Prempro case goes to trial. Looks like major news outlets are now reporting what was posted herein February of 2006 on the first trial, which you can read here.
From cnn.com:
"The overwhelming majority of women in these cases suffer from breast cancer as a result of their use of Prempro," said lawyer Tobias Millrood, whose firm Schiffrin & Barroway is representing plaintiffs in about 1,000 Prempro cases. "Wyeth aggressively over-promoted these drugs for conditions that [weren't approved by] the FDA. [Prempro] was truly a panacea. This drug was promoted as the fountain of youth."
The first case is set to start on July 31 in federal district court in Little Rock, Ark. Plaintiff Linda Reeves, 67, of Benton, Ark., was diagnosed with breast cancer after taking Prempro for eight years for the prevention of osteoporosis, according to her lawyer, Jim Morris, also of Schiffrin & Barroway. Reeves blames Prempro for the cancer that led to her mastectomy.
The second Prempro case is set for September 11, 2006 in state court in Philadelphia. The Plaintiff is a 67-year-old woman from Dayton, Ohio, who blames her six years of Prempro use for causing the cancer that led to her double mastectomy in 2001.
In June, the first Prempro case goes to trial. Looks like major news outlets are now reporting what was posted herein February of 2006 on the first trial, which you can read here.
From cnn.com:
"The overwhelming majority of women in these cases suffer from breast cancer as a result of their use of Prempro," said lawyer Tobias Millrood, whose firm Schiffrin & Barroway is representing plaintiffs in about 1,000 Prempro cases. "Wyeth aggressively over-promoted these drugs for conditions that [weren't approved by] the FDA. [Prempro] was truly a panacea. This drug was promoted as the fountain of youth."
The first case is set to start on July 31 in federal district court in Little Rock, Ark. Plaintiff Linda Reeves, 67, of Benton, Ark., was diagnosed with breast cancer after taking Prempro for eight years for the prevention of osteoporosis, according to her lawyer, Jim Morris, also of Schiffrin & Barroway. Reeves blames Prempro for the cancer that led to her mastectomy.
The second Prempro case is set for September 11, 2006 in state court in Philadelphia. The Plaintiff is a 67-year-old woman from Dayton, Ohio, who blames her six years of Prempro use for causing the cancer that led to her double mastectomy in 2001.
Wednesday, April 26, 2006
Canada and the Infected Tissue/Bone Scandal
It is not just in Oklahoma City, or Atlanta, GA or on Long Island. News today shows that this is a problem for all of North America:
A Vancouver woman who found out that she may have received infected donor tissue during reconstructive eardrum surgery in 1994 is proceeding with what she thinks may be a class-action lawsuit against St. Paul's Hospital, the B.C. Ear Bank and a number of other defendants.
The lawsuit follows a Health Canada order three years ago to recall all unused tissue and bone distributed by the B.C. Ear Bank and notify all recipient institutions.
The scandal was reported by The Vancouver Sun in 2003. It was noted that St. Paul's Hospital officials acknowledged problems with the record-keeping and day-to-day operations of the B.C. Ear Bank, which had been housed in the hospital since 1995 but was shut down in 2002.
The ear bank, which collected, processed and stored bone and tissue, distributed 6,016 specimens to about 80 hospitals across North America for transplant and research purposes.
Source: Canada.com
A Vancouver woman who found out that she may have received infected donor tissue during reconstructive eardrum surgery in 1994 is proceeding with what she thinks may be a class-action lawsuit against St. Paul's Hospital, the B.C. Ear Bank and a number of other defendants.
The lawsuit follows a Health Canada order three years ago to recall all unused tissue and bone distributed by the B.C. Ear Bank and notify all recipient institutions.
The scandal was reported by The Vancouver Sun in 2003. It was noted that St. Paul's Hospital officials acknowledged problems with the record-keeping and day-to-day operations of the B.C. Ear Bank, which had been housed in the hospital since 1995 but was shut down in 2002.
The ear bank, which collected, processed and stored bone and tissue, distributed 6,016 specimens to about 80 hospitals across North America for transplant and research purposes.
Source: Canada.com
Merck Article in Fortune
Good stuff on Fortune.com regarding Merck's "fight every case" strategy:
"Merck's defense of Vioxx is beginning to look ugly - and costing the company millions in legal fees and punitive damages. But make no mistake, the company's fight-every-case strategy is still its only rational option ...
For those of you keeping score at home, Merck has some 11,500 more cases to fight. So far, the company has faced six plaintiffs (in five trials) and prevailed against three of them. They're playing .500 ball.
In spite of some high-dollar jury awards, the company's general counsel, Ken Frazier says he's satisfied with the better-than-expected win-loss tally.
The key question before juries is: Did Vioxx cause this particular plaintiff's heart attack? So far, some jurors show a willingness to hear both sides and eliminate plaintiffs who appear to have invalid claims.
That's exactly the kind of consideration Merck needs. Despite what they say, Merck's lawyers don't really intend to fight every one of these 11,000 cases. The company's current strategy is a temporary one.
To most legal experts, it's the only legal strategy that makes sense. "What's the alternative?" asks Andrew Kaufman, a professor of law at Harvard University. "If you establish a settlement fund at this point, you're asking more people to file suit. A settlement in the absense of trials means inflated costs."
The only scenario in which they won't settle is if they win so much that all the plaintiffs go away. Based on the record so far, that isn't likely to happen."
"Merck's defense of Vioxx is beginning to look ugly - and costing the company millions in legal fees and punitive damages. But make no mistake, the company's fight-every-case strategy is still its only rational option ...
For those of you keeping score at home, Merck has some 11,500 more cases to fight. So far, the company has faced six plaintiffs (in five trials) and prevailed against three of them. They're playing .500 ball.
In spite of some high-dollar jury awards, the company's general counsel, Ken Frazier says he's satisfied with the better-than-expected win-loss tally.
The key question before juries is: Did Vioxx cause this particular plaintiff's heart attack? So far, some jurors show a willingness to hear both sides and eliminate plaintiffs who appear to have invalid claims.
That's exactly the kind of consideration Merck needs. Despite what they say, Merck's lawyers don't really intend to fight every one of these 11,000 cases. The company's current strategy is a temporary one.
To most legal experts, it's the only legal strategy that makes sense. "What's the alternative?" asks Andrew Kaufman, a professor of law at Harvard University. "If you establish a settlement fund at this point, you're asking more people to file suit. A settlement in the absense of trials means inflated costs."
The only scenario in which they won't settle is if they win so much that all the plaintiffs go away. Based on the record so far, that isn't likely to happen."
Monday, April 24, 2006
Garza Vioxx Verdict: Trial Analysis
This case was tried only one week per month and as a result not easy to track. A member of the trial team provided the analysis below:
Leonel Garza was a 71 year old smoker with an extensive cardiac history - prior heart attack, bypass, stent placement. Retired military who worked as assistant County Auditor until he retired from that as well. Very active and careful since had previous heart problems. His wife testified that he had cut down his smoking from two packs a day to 3-4 cigarettes a day. Hypertension, hyperlipidemia, treated and well controlled.
Garza visited a cardiologist 3/27/01 complaining of arm pain. Doctor placed him on a seven day trial of an "anti-inflammatory" Only reference to Vioxx was a note made on billing sheet. Had a thallium scan performed which showed good perfusionand only mild abnormality in the region of previous mi. Dr Juan Posada (not his regular cardiologist) saw him in follow- up on 4/4/01 as Dr Evans was out. His notes reflect that he had arthritis symptoms which had "improved markedly" (because of the Vioxx)
Wife testified that Dr. Posada provided samples until his next visit (in a month) with Evans. His records reflected that he offered the "possibility" of a cath but garza wanted to discuss it with Evans. Posada agreed with the decision based on the absence of symptoms. Although Posada's records do not reflect that samples were provided, he testified that it was possible. Additionally, he said that it was more likely than not that a reasonable and prudent physician would continue medication that provided pain relief. Records also reflect that he was on 'optimal medical management"
On April 21, 2001, he was found dead beside his truck at his ranch. Autopsy revealed two fresh thrombotic occlusions. Case tried by David Hockema and Joe Escobedo from the firm of Hockema Tippett and Escobedo of Edinburg, and Kathy Snapka Turman& Waterhouse of Corpus Christi. Ms. Snapka provided this analysis.
Leonel Garza was a 71 year old smoker with an extensive cardiac history - prior heart attack, bypass, stent placement. Retired military who worked as assistant County Auditor until he retired from that as well. Very active and careful since had previous heart problems. His wife testified that he had cut down his smoking from two packs a day to 3-4 cigarettes a day. Hypertension, hyperlipidemia, treated and well controlled.
Garza visited a cardiologist 3/27/01 complaining of arm pain. Doctor placed him on a seven day trial of an "anti-inflammatory" Only reference to Vioxx was a note made on billing sheet. Had a thallium scan performed which showed good perfusionand only mild abnormality in the region of previous mi. Dr Juan Posada (not his regular cardiologist) saw him in follow- up on 4/4/01 as Dr Evans was out. His notes reflect that he had arthritis symptoms which had "improved markedly" (because of the Vioxx)
Wife testified that Dr. Posada provided samples until his next visit (in a month) with Evans. His records reflected that he offered the "possibility" of a cath but garza wanted to discuss it with Evans. Posada agreed with the decision based on the absence of symptoms. Although Posada's records do not reflect that samples were provided, he testified that it was possible. Additionally, he said that it was more likely than not that a reasonable and prudent physician would continue medication that provided pain relief. Records also reflect that he was on 'optimal medical management"
On April 21, 2001, he was found dead beside his truck at his ranch. Autopsy revealed two fresh thrombotic occlusions. Case tried by David Hockema and Joe Escobedo from the firm of Hockema Tippett and Escobedo of Edinburg, and Kathy Snapka Turman& Waterhouse of Corpus Christi. Ms. Snapka provided this analysis.
Saturday, April 22, 2006
Bonescandal.com: Infected Bone and Tissue Site
We have put together a website that details the widening contaminated/infected bone and tissue scandal. Also this weekend, you can read about the Tainted Body Parts scandal in the south at ajc.com.
You can find it by going to www.bonescandal.com. It details the potential or alleged wrongdoers and provides information on what is happening now.
You can find it by going to www.bonescandal.com. It details the potential or alleged wrongdoers and provides information on what is happening now.
Texas Garza Vioxx Verdict: For Plaintiff
$32 million for the Estate, punitives awarded. More information to follow. Most who followed the case are surprised with the verdict.
Friday, April 21, 2006
Bone and Tissue Scandal: News hits in the U.K., Texas & OK
Recent articles in the Oklahoma City, OK newspaper as well as articles in Austin, Texas, Long Island, NY and Iowa newspapers tend to be more straightforward when detailing the infected/contaminated tissue and bone scandal. Not so in the U.K.
From the Observer's online edition, you can read the story that includes this text:
Every graveyard tells a story. The Maple Grove Cemetery in New York is no exception. Its tombs mirror the waves of immigration to this bustling corner of the city. Inscriptions on older stones, flecked with moss, are mostly Jewish or English. The newer ones, shiny and polished, bear Chinese names, or Indian, or Hispanic. Each grave is a full stop on an individual life: a point of final departure and no return. Except for one.
You can read more by going here.
You can also go to www.bonescandal.com for more information.
From the Observer's online edition, you can read the story that includes this text:
Every graveyard tells a story. The Maple Grove Cemetery in New York is no exception. Its tombs mirror the waves of immigration to this bustling corner of the city. Inscriptions on older stones, flecked with moss, are mostly Jewish or English. The newer ones, shiny and polished, bear Chinese names, or Indian, or Hispanic. Each grave is a full stop on an individual life: a point of final departure and no return. Except for one.
You can read more by going here.
You can also go to www.bonescandal.com for more information.
Thursday, April 20, 2006
Garza Trial: Attorney asks for $1B +
I just shake my head at tactcic like this. From Reuters:
In the Garza trial, the damage request includes $22 million for mental anguish and personal loss and $1 billion in punitive damages.
As was noted in my earlier post, punitive damages in that case have been capped at $750,000 under Texas law, meaning the Plaintiffs could collect no more than that even if the jury awarded the full $1 billion.
In the Garza trial, the damage request includes $22 million for mental anguish and personal loss and $1 billion in punitive damages.
As was noted in my earlier post, punitive damages in that case have been capped at $750,000 under Texas law, meaning the Plaintiffs could collect no more than that even if the jury awarded the full $1 billion.
Florida: Battle rages over PIP
From various sources including the Miami Herald, the AFTL, the Bradenton paper, and the PB Post:
The Senate's no-fault auto insurance bill is picking up some steam, but it does not include many of the measures that insurers want. By way of background, the bill would extend the state's no-fault law to 2009, which is now set to expire in October. The designated October date is the "sunset" date.
On the House side, a companion bill will be heard today that extends Personal Injury Protection to 2012.
The bill gives $2.6 million to add 19 positions to the Department of Financial Services' Division of Insurance Fraud and provide pay raises for existing personnel. It also adds $750,000 to pay for six new assistant state attorneys to work solely on insurance fraud cases.
Florida is one of twelve states with no-fault auto insurance laws. Each driver must buy at least $10,000 in PIP coverage, which guarantees medical, disability and death benefits. In return, accident victims give up the right to sue for pain and suffering unless they have sustained a permanent injury.
For their part, most healthcare providers and attorneys would like to see the current law extended with no changes.
I'm licensd in Florida. When PIP-type coverage was done away with in Georgia the battle cry from the insurers was 'lower premiums' but that of course never materialized.
In the end, unpaid bills from auto wrecks were paid by ... guess who? If you said "taxpayers" you would be right. Many bills are now submitted to Georgia Medicaid which pays them and in return seeks some level of subrogation.
The Senate's no-fault auto insurance bill is picking up some steam, but it does not include many of the measures that insurers want. By way of background, the bill would extend the state's no-fault law to 2009, which is now set to expire in October. The designated October date is the "sunset" date.
On the House side, a companion bill will be heard today that extends Personal Injury Protection to 2012.
The bill gives $2.6 million to add 19 positions to the Department of Financial Services' Division of Insurance Fraud and provide pay raises for existing personnel. It also adds $750,000 to pay for six new assistant state attorneys to work solely on insurance fraud cases.
Florida is one of twelve states with no-fault auto insurance laws. Each driver must buy at least $10,000 in PIP coverage, which guarantees medical, disability and death benefits. In return, accident victims give up the right to sue for pain and suffering unless they have sustained a permanent injury.
For their part, most healthcare providers and attorneys would like to see the current law extended with no changes.
I'm licensd in Florida. When PIP-type coverage was done away with in Georgia the battle cry from the insurers was 'lower premiums' but that of course never materialized.
In the end, unpaid bills from auto wrecks were paid by ... guess who? If you said "taxpayers" you would be right. Many bills are now submitted to Georgia Medicaid which pays them and in return seeks some level of subrogation.
A Post Worth Reading: Duct Tape Marketing
John Jantsch at Duct Tape Marketing has a blog worth reading. Last week he wrote: Your Marketing Stinks and It's Your Fault.
While he writes for business, you may find his posts helpful if you run a law firm. He wrote last week:
The reason so many small businesses struggle is because they are unwilling to change. The marketing strategy, if there is one, is to do what everyone else in the industry seems to be doing. If that's your marketing reality, that little safe place, and it's not working, the first thing you have to come to grips with is that it is your fault... The only marketing strategy you need to worry about as a small business is to find a way to be unique.
Would this apply to law? It's worth looking to see what niche may work. The key is sticking to any marketing plan. For my law practice in Georgia one key component to our marketing plan: When it comes to a specific type of referral from a fellow lawyer, I pay that attorney/firm 50% of any legal fees derived, and when a case is successfully concluded, the client receives a letter directing him or her back to the originating firm. I don't keep that client in our database for any marketing purposes.
The problem I see is not so much having a unique marketing plan. The plan 'stinks' - fails - in the law business when there is insufficient execution of the plan.
While he writes for business, you may find his posts helpful if you run a law firm. He wrote last week:
The reason so many small businesses struggle is because they are unwilling to change. The marketing strategy, if there is one, is to do what everyone else in the industry seems to be doing. If that's your marketing reality, that little safe place, and it's not working, the first thing you have to come to grips with is that it is your fault... The only marketing strategy you need to worry about as a small business is to find a way to be unique.
Would this apply to law? It's worth looking to see what niche may work. The key is sticking to any marketing plan. For my law practice in Georgia one key component to our marketing plan: When it comes to a specific type of referral from a fellow lawyer, I pay that attorney/firm 50% of any legal fees derived, and when a case is successfully concluded, the client receives a letter directing him or her back to the originating firm. I don't keep that client in our database for any marketing purposes.
The problem I see is not so much having a unique marketing plan. The plan 'stinks' - fails - in the law business when there is insufficient execution of the plan.
Vioxx: Garza Trial (TX) Damages Capped
In the Texas State Court case involving Garza (a short term use case), yesterday the Court refused a Plaintiffs' request to ask jurors to decide during deliberations whether Merck had criminally caused the death of an elderly person.
What does the ruling mean? Merck, under Texas law, will now face at most a $750,000 capped award for punitive damages (if any). A ruling the other way would have taken all limits off.
The Plaintiff is Leonel Garza of Rio Grande City. He died of a heart attack in 2001 after taking Vioxx for no more than 24 days for arm pain. Merck has introduced evidence showing a 20+ year history of heart disease.
Sources:
This is from Reuters, Yahoo, the local TX papers.
What does the ruling mean? Merck, under Texas law, will now face at most a $750,000 capped award for punitive damages (if any). A ruling the other way would have taken all limits off.
The Plaintiff is Leonel Garza of Rio Grande City. He died of a heart attack in 2001 after taking Vioxx for no more than 24 days for arm pain. Merck has introduced evidence showing a 20+ year history of heart disease.
Sources:
This is from Reuters, Yahoo, the local TX papers.
Wednesday, April 19, 2006
Tequin (An Antiobiotic) News: Link to Diabetes?
Tequin (gatifloxacin), an antiobiotic is that widely prescribed, may be linked to diabetes and other blood sugar conditions and disorders.
The New England Journal of Medicine will publish a report shortly that shows Tequin users had 17 times greater risk of developing serious diabetes and 4 times greater risk of being hospitalized with low blood sugar complications than patients using other antibiotics.
Last month, the Canadian study released linked Tequin to diabetes and other blood sugar disorders. Dr. David Juurlink of the Sunnybrook and Women's College Health Sciences Center in Toronto asked the health industry if it would be perhaps in consumers' best interests to pull the drug. Go here to read the NEJM article in summary form.
This year FDA added a warning to the Tequin label. What is significant however is the report that problems can arise from short term usage of the medication.
The New England Journal of Medicine will publish a report shortly that shows Tequin users had 17 times greater risk of developing serious diabetes and 4 times greater risk of being hospitalized with low blood sugar complications than patients using other antibiotics.
Last month, the Canadian study released linked Tequin to diabetes and other blood sugar disorders. Dr. David Juurlink of the Sunnybrook and Women's College Health Sciences Center in Toronto asked the health industry if it would be perhaps in consumers' best interests to pull the drug. Go here to read the NEJM article in summary form.
This year FDA added a warning to the Tequin label. What is significant however is the report that problems can arise from short term usage of the medication.
NJ Vioxx Court Update, April 2006
From a recent case management conference:
Judge Higbee is concerned about a mass filing of cases on September 30,2006.
The Judge would prefer that cases a filed in smaller batches,
periodically between now and the September 30, 2006 SOL deadline.
The Court has proposed several measures to expedite future Vioxx
trials, including providing juries with: stipulated calendars and
summarizing key dates.
The Court has expressed interest in addressing stroke and GI-injury
cases on file.
The Court denied Merck's motion to stay the NJ litigation pending
appellate review of the Cona/McDarby decisions.
Judge Higbee is concerned about a mass filing of cases on September 30,2006.
The Judge would prefer that cases a filed in smaller batches,
periodically between now and the September 30, 2006 SOL deadline.
The Court has proposed several measures to expedite future Vioxx
trials, including providing juries with: stipulated calendars and
summarizing key dates.
The Court has expressed interest in addressing stroke and GI-injury
cases on file.
The Court denied Merck's motion to stay the NJ litigation pending
appellate review of the Cona/McDarby decisions.
Bone and Tissue Scandal: Funeral Home Raided in Philly
From various sources including the Philadelphia paper online:
The D.A.'s office raided a funeral home located in Kensington, PA. The reason? Interviews are being conducted in an attempt to obtain informtino of the home in the developign body-parts scandal.
Philly investigators have talked to at least two former techs from Biomedical Tissue Services Inc. ("BTS"). Several now allege BTS removed bones, veins and tendons from corpses of deceased Philadelphians, according to sources close to the investigation. (Per the Philly papers)
The D.A.'s office is investigating how many Philly corpses were dissected, how much money was exchanged for the work, and how several funeral homes began a contractual relationship with BTS.
In PA, only corneas can be taken from corpses inside funeral homes.
You can read more at www.bonescandal.com. I know that my friend Steve Gacovino is also on the web at infectedtissueandbone.com.
The D.A.'s office raided a funeral home located in Kensington, PA. The reason? Interviews are being conducted in an attempt to obtain informtino of the home in the developign body-parts scandal.
Philly investigators have talked to at least two former techs from Biomedical Tissue Services Inc. ("BTS"). Several now allege BTS removed bones, veins and tendons from corpses of deceased Philadelphians, according to sources close to the investigation. (Per the Philly papers)
The D.A.'s office is investigating how many Philly corpses were dissected, how much money was exchanged for the work, and how several funeral homes began a contractual relationship with BTS.
In PA, only corneas can be taken from corpses inside funeral homes.
You can read more at www.bonescandal.com. I know that my friend Steve Gacovino is also on the web at infectedtissueandbone.com.
Tuesday, April 18, 2006
Tech This N' That: Shorttext.com
Shorttext.com is a site where you can post any information and share it with others. Post your text below and we will save it and generate a URL for you. This URL is for you to distribute, print, post, so says that site.
It's a nifty little site that can help in a pinch. Today I was in another's office, and found a site that I thought I could use later. I didn't want to log in to my email, or have someone come over to the PC to show me a way to email the site. I went to shorttext.com, pasted the link, and emailed it from that site. It creates a URL to be emailed. Know that there are public and private URL linke you can create.
Hope you put the site to good use. Did I mention the site is free?
It's a nifty little site that can help in a pinch. Today I was in another's office, and found a site that I thought I could use later. I didn't want to log in to my email, or have someone come over to the PC to show me a way to email the site. I went to shorttext.com, pasted the link, and emailed it from that site. It creates a URL to be emailed. Know that there are public and private URL linke you can create.
Hope you put the site to good use. Did I mention the site is free?
Fosamax News (Bone Loss Drug) - Merck's Next Litigation Problem?
Fosamax made the news last week.
Fosamax is a drug indicated for the treatment and prevention of osteoporosis (thinning of the bones) in men and women. A study recently linked the use of bisphosphonates, such as Fosamax with osteonecrosis of the jaw. It is frequently referred to as "jaw death." Merck makes this drug.
Osteonecrosis is a bone disease that few have heard of. Somewhere between 10,000 and 20,000 people are diagnosed with this disease each year in the United States alone. You can read more about the condition here.
The condition is associated with the interference of the blood supply to the bone and the consequential damage that occurs. Osteonecrosis of the jaw can be painful and could lead to infection, breakdown of the jawbone, and ulcerations in the lining of the mouth.
From the Online Journal:
The osteoporosis drug Fosamax has been on the market for a little over 10 years now. Drug maker Merck promoted it heavily by selling women the fear of a disabling hip fracture and the necessity of regular bone-density tests. Merck's initial TV advertising campaign featured a slim woman in her mid-40s, conveying the notion that testing was appropriate for women in this age group.
n a 2004 letter published in the Annals of Internal Medicine, researcher Susan Ott, MD, of the University of Washington wrote: "Many people believe that these drugs are 'bone builders,' but the evidence shows they are actually bone hardeners."
In a December 13, 2004, press release, doctors at Long Island Jewish (LIJ) Medical Center announced that they had discovered a link between a common chemotherapy drug and a serious bone disease called osteonecrosis of the jaw (ONJ).
The discovery, published in the Journal of Oral and Maxillofacial Surgeons, prompted both the FDA and Novartis to issue warnings to physicians and dentists about the risk for the potential adverse effect.
You can read more about the condition at that site.
Fosamax is a drug indicated for the treatment and prevention of osteoporosis (thinning of the bones) in men and women. A study recently linked the use of bisphosphonates, such as Fosamax with osteonecrosis of the jaw. It is frequently referred to as "jaw death." Merck makes this drug.
Osteonecrosis is a bone disease that few have heard of. Somewhere between 10,000 and 20,000 people are diagnosed with this disease each year in the United States alone. You can read more about the condition here.
The condition is associated with the interference of the blood supply to the bone and the consequential damage that occurs. Osteonecrosis of the jaw can be painful and could lead to infection, breakdown of the jawbone, and ulcerations in the lining of the mouth.
From the Online Journal:
The osteoporosis drug Fosamax has been on the market for a little over 10 years now. Drug maker Merck promoted it heavily by selling women the fear of a disabling hip fracture and the necessity of regular bone-density tests. Merck's initial TV advertising campaign featured a slim woman in her mid-40s, conveying the notion that testing was appropriate for women in this age group.
n a 2004 letter published in the Annals of Internal Medicine, researcher Susan Ott, MD, of the University of Washington wrote: "Many people believe that these drugs are 'bone builders,' but the evidence shows they are actually bone hardeners."
In a December 13, 2004, press release, doctors at Long Island Jewish (LIJ) Medical Center announced that they had discovered a link between a common chemotherapy drug and a serious bone disease called osteonecrosis of the jaw (ONJ).
The discovery, published in the Journal of Oral and Maxillofacial Surgeons, prompted both the FDA and Novartis to issue warnings to physicians and dentists about the risk for the potential adverse effect.
You can read more about the condition at that site.
Thursday, April 13, 2006
Vioxx Verdict in NJ: A Juror speaks
From AP, Newsday.com, and Yahoo!:
Carla Tennyson was one of the jurors in the McDarby case. She has been reported as saying, that Merck has "an obligation to tell us the facts and let us decide if the risk was worth taking," she said. "That's all it is. For me, it was right from the beginning the label. They could've gotten a new label in 30 days, as soon as they realized there was a problem. They didn't."
Also, it should be noted that in New Jersey the punitive damages award might result in a criminal probe. That state requires that punitive damages cases be referred to the county prosecutor and the state Attorney General's Office for investigation into whether a criminal act was committed.
The statute is 2A:15-5.17. "Record referred for criminal investigation" and provides "Upon the conclusion of any action in which punitive damages have been awarded, the court shall refer the record of that action to the prosecutor of the county in which the case was tried and to the Attorney General for investigation as to whether a criminal act has been committed by the defendant."
Carla Tennyson was one of the jurors in the McDarby case. She has been reported as saying, that Merck has "an obligation to tell us the facts and let us decide if the risk was worth taking," she said. "That's all it is. For me, it was right from the beginning the label. They could've gotten a new label in 30 days, as soon as they realized there was a problem. They didn't."
Also, it should be noted that in New Jersey the punitive damages award might result in a criminal probe. That state requires that punitive damages cases be referred to the county prosecutor and the state Attorney General's Office for investigation into whether a criminal act was committed.
The statute is 2A:15-5.17. "Record referred for criminal investigation" and provides "Upon the conclusion of any action in which punitive damages have been awarded, the court shall refer the record of that action to the prosecutor of the county in which the case was tried and to the Attorney General for investigation as to whether a criminal act has been committed by the defendant."
Tuesday, April 11, 2006
The Widening Scandal: Contaminated Bone and Tissue Implants
My office is investigating what to me are some of the most disturbing claims I have run across as an attorney. Patients have had implanted in them without their knowledge infected or contaminated bone and tissue parts while undergoing surgery for a back injury, a dental injury, or a fusion. Good hardworking people are now suddenly getting sick. Some have been diagnosed as having Hepatitis B or C, or worse. I wanted to jot down in a longer form what is happening to these folks to alert counsel across the country.
Bone and Tissue for Sale- How this scandal began
The practice of illegal trafficking in human organs, body parts, and tissue has made the news lately, and people have been indicted over the practice. Paitents who had bone or tissue implants placed in them without their knowledge are now getting sick and developing diseases like Hepatitis B and worse.
While the scandal of stealing bones from cadavers being falsely certified as clean continues to erupt in the headlines and the courtrooms, where did this mess all start? I can trace it back to UCLA’s Director of the Willed Body Program, Henry Reid. Reid was arrested and a criminal investigation launched several years ago.
The UCLA Medical Center investigation started in 1998. In March of 2004 Ernest Nelson was arrested - he claimed to have paid Director Reid hundreds of thousands of dollars for permission to enter the UCLA body freezer and and harvest body parts from hundreds of cadavers. You can read more here
about the arrest. The Los Angeles Times reported in 2004 that it was shown invoices on a UCLA letterhead indicating 496 cadavers were sold for a total of $704,600 between 1998 and 2003. One client was pharmaceutical giant Johnson & Johnson. Go here for the article.
Companies in the medical industry pay for a wide range of body parts including skin, scalps, teeth, skulls and bones, which are then used for among other things surgery. Heart valves can fetch $5,000 to $7,000 each. Skin, used to dress the wounds of burn victims, can go for $1,000 a square foot.If a single body could provide all its organs, the cadaver would be worth $150,000.
In 2004, Johnson & Johnson was named in court documents as having contracted with Nelson for human tissue. Later, a California law firm filed a class action.
The quest to have a class certified in that state failed. The California Court of Appeal issued a decision that may materially affect the ability to certify the matter. In Bennett v. The Regents of the University of California, 133 Cal.App.4th 347 (2005), the Court of Appeals, in dicta, implied that cases involving the mishandling of human remains do not generally lend themselves to class certification.
The Risk:
One of the risks associated with the illegal harvesting or snatching of body parts is that the ones who do it avoid screening and testing procedures set up and maintained to ensure recipients will not receive diseased or contaminated tissue, bone, or organs. With the possibility that many patients could receive tissue or bone from a single infected cadaver, the potential for a medical catastrophe cannot be minimized.
The FDA has issued reports on the harvesting, and in December of 2005 wrote on its website:
The Food and Drug Administration (FDA) is notifying the public of its investigation of human tissue recovered by Biomedical Tissue Services, Ltd (BTS) of Fort Lee, NJ and sent to tissue processors. Some of this tissue (e.g., human bone, skin, tendons) may have been implanted into patients from early 2004 to September 2005. The tissue was recovered by BTS from human donors who may not have met FDA donor eligibility requirements and who may not have been properly screened for certain infectious diseases.
Of special interest to periodontists are LifeCell Corporation, which produces Alloderm for BioHorizons, Inc., and Totugen Medical Inc, which produces Puros for Zimmer Dental. No adverse reactions related to these tissues have been reported at this time and the FDA and the Centers for Disease Control (CDC) believe the risk of infection from these tissues is low. It is important to determine whether the allograft material is from the recalled lots. Dentists and physicians who have obtained tissue affected by the recall should have been notified by now
You can find that information by going here. This news was preceeded by an October 2005 alert titled: FDA Provides Information on Investigation into Human Tissue for Transplantation, which can be found here.
In November, Medtronic informed the FDA that it had tracked 13,000 contaminated samples, and had 8,000 implanted.
News in 2006
In Brooklyn New York, more than one thousand bodies are part of the District Attorney’s investigations into the theft and sale of bones and other body parts removed from fresh corpses at several funeral homes, without permission. These were then sold to BioMedical Tissue Services, a Fort Lee, N.J., tissue recovery company run by Michael Mastromarino. There have been indictments issued, and they were handed down in February of this year. Go here for more information. The prosecutor has been quoted as saying that the case was "like something out of a cheap horror movie." Bones were replaced with everything from broomsticks and pipes to plumbing supplies. An AP photo can be found below showing just that:
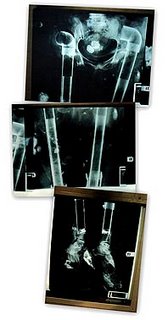
BTS is not alone. Lifecell Inc. has issued a recall of certain products made from body parts acquired from BioMedical Tissue Services. They are AlloDerm, used for plastic surgery, burn and periodontal procedures; Repliform used for gynecological and urological surgical procedures; and GraftJacket, used for orthopedic applications and lower extremity wounds.
The Philadelphia Inquirer reported last month that more indictments are expected.
In addition, many medical facilities and hospitals have directly written to patients to notify them of the possibility that they may have contracted any one or more of a number of serious and even life-threatening diseases from the bone or tissue grafts they received.
For example, The North Shore-Long Island Jewish Health System has mailed letters to former patients alerting them that they may have been exposed to potentially contaminated body parts. The letters state the hospital had indirectly received human bone, skin and tendons from BioMedical Tissue Services, which may not have properly screened the items for infectious diseases.
Health officials are concerned that tens of thousands of people across the country, and possibly more on Long Island, may have been exposed by untested parts from BTS
BioMedical is already being sued by many who claim a relative’s body parts were stolen from the grave and sold to the New Jersey company. Hundreds are already being tested for various diseases. BTS supplied bone, skin and tendons to various processors, who in turn provided them to distributors.
From the news developing, many of the body parts used on Long Island were purchased from BioMedical by a Florida tissue bank responsible for testing and sterilizing every body part it buys. Regeneration Technologies, based in Gainesville has or will be inmplicated.
Based upon the revelations so far, the scandal involving body parts, tissue, and bones promises to be major news for the next eighteen months or more.
Sources include:
nypress.comas well as articles on newsinferno and the daily news, and the FDA or at www.bonescandal.com
Bone and Tissue for Sale- How this scandal began
The practice of illegal trafficking in human organs, body parts, and tissue has made the news lately, and people have been indicted over the practice. Paitents who had bone or tissue implants placed in them without their knowledge are now getting sick and developing diseases like Hepatitis B and worse.
While the scandal of stealing bones from cadavers being falsely certified as clean continues to erupt in the headlines and the courtrooms, where did this mess all start? I can trace it back to UCLA’s Director of the Willed Body Program, Henry Reid. Reid was arrested and a criminal investigation launched several years ago.
The UCLA Medical Center investigation started in 1998. In March of 2004 Ernest Nelson was arrested - he claimed to have paid Director Reid hundreds of thousands of dollars for permission to enter the UCLA body freezer and and harvest body parts from hundreds of cadavers. You can read more here
about the arrest. The Los Angeles Times reported in 2004 that it was shown invoices on a UCLA letterhead indicating 496 cadavers were sold for a total of $704,600 between 1998 and 2003. One client was pharmaceutical giant Johnson & Johnson. Go here for the article.
Companies in the medical industry pay for a wide range of body parts including skin, scalps, teeth, skulls and bones, which are then used for among other things surgery. Heart valves can fetch $5,000 to $7,000 each. Skin, used to dress the wounds of burn victims, can go for $1,000 a square foot.If a single body could provide all its organs, the cadaver would be worth $150,000.
In 2004, Johnson & Johnson was named in court documents as having contracted with Nelson for human tissue. Later, a California law firm filed a class action.
The quest to have a class certified in that state failed. The California Court of Appeal issued a decision that may materially affect the ability to certify the matter. In Bennett v. The Regents of the University of California, 133 Cal.App.4th 347 (2005), the Court of Appeals, in dicta, implied that cases involving the mishandling of human remains do not generally lend themselves to class certification.
The Risk:
One of the risks associated with the illegal harvesting or snatching of body parts is that the ones who do it avoid screening and testing procedures set up and maintained to ensure recipients will not receive diseased or contaminated tissue, bone, or organs. With the possibility that many patients could receive tissue or bone from a single infected cadaver, the potential for a medical catastrophe cannot be minimized.
The FDA has issued reports on the harvesting, and in December of 2005 wrote on its website:
The Food and Drug Administration (FDA) is notifying the public of its investigation of human tissue recovered by Biomedical Tissue Services, Ltd (BTS) of Fort Lee, NJ and sent to tissue processors. Some of this tissue (e.g., human bone, skin, tendons) may have been implanted into patients from early 2004 to September 2005. The tissue was recovered by BTS from human donors who may not have met FDA donor eligibility requirements and who may not have been properly screened for certain infectious diseases.
Of special interest to periodontists are LifeCell Corporation, which produces Alloderm for BioHorizons, Inc., and Totugen Medical Inc, which produces Puros for Zimmer Dental. No adverse reactions related to these tissues have been reported at this time and the FDA and the Centers for Disease Control (CDC) believe the risk of infection from these tissues is low. It is important to determine whether the allograft material is from the recalled lots. Dentists and physicians who have obtained tissue affected by the recall should have been notified by now
You can find that information by going here. This news was preceeded by an October 2005 alert titled: FDA Provides Information on Investigation into Human Tissue for Transplantation, which can be found here.
In November, Medtronic informed the FDA that it had tracked 13,000 contaminated samples, and had 8,000 implanted.
News in 2006
In Brooklyn New York, more than one thousand bodies are part of the District Attorney’s investigations into the theft and sale of bones and other body parts removed from fresh corpses at several funeral homes, without permission. These were then sold to BioMedical Tissue Services, a Fort Lee, N.J., tissue recovery company run by Michael Mastromarino. There have been indictments issued, and they were handed down in February of this year. Go here for more information. The prosecutor has been quoted as saying that the case was "like something out of a cheap horror movie." Bones were replaced with everything from broomsticks and pipes to plumbing supplies. An AP photo can be found below showing just that:

BTS is not alone. Lifecell Inc. has issued a recall of certain products made from body parts acquired from BioMedical Tissue Services. They are AlloDerm, used for plastic surgery, burn and periodontal procedures; Repliform used for gynecological and urological surgical procedures; and GraftJacket, used for orthopedic applications and lower extremity wounds.
The Philadelphia Inquirer reported last month that more indictments are expected.
In addition, many medical facilities and hospitals have directly written to patients to notify them of the possibility that they may have contracted any one or more of a number of serious and even life-threatening diseases from the bone or tissue grafts they received.
For example, The North Shore-Long Island Jewish Health System has mailed letters to former patients alerting them that they may have been exposed to potentially contaminated body parts. The letters state the hospital had indirectly received human bone, skin and tendons from BioMedical Tissue Services, which may not have properly screened the items for infectious diseases.
Health officials are concerned that tens of thousands of people across the country, and possibly more on Long Island, may have been exposed by untested parts from BTS
BioMedical is already being sued by many who claim a relative’s body parts were stolen from the grave and sold to the New Jersey company. Hundreds are already being tested for various diseases. BTS supplied bone, skin and tendons to various processors, who in turn provided them to distributors.
From the news developing, many of the body parts used on Long Island were purchased from BioMedical by a Florida tissue bank responsible for testing and sterilizing every body part it buys. Regeneration Technologies, based in Gainesville has or will be inmplicated.
Based upon the revelations so far, the scandal involving body parts, tissue, and bones promises to be major news for the next eighteen months or more.
Sources include:
nypress.comas well as articles on newsinferno and the daily news, and the FDA or at www.bonescandal.com
Print from your computer to any Kinko's anywhere
"Print anywhere in the world" is the title of a post you can find at The Practice Blog. When I'm on a trip I carry a list of Kinko's that are near my hotel or meeting, because you just never know when a web connection will fail, or you need something that you may have forgotten. This post to me is golden, and I repost it in full ...
This link will bring you to Kinkos. You can download and install software. When you are ready to print a document, you click on this, and off it goes. You can have it bound, hole punched, or enlarged.
So many uses for this. You can print a document to the Kinko's nearest a client and then the client can just pick it up. A virtual law clerk could research and write a memo and then have the attorney pick up a printed version. Law students could use this to print out papers and save the hassle of printing it at home, especially late at night the day before it is due when printers have a habit of breaking.
This is a great little program that you can use.
Many thanks, Jonathan and Terry!
This link will bring you to Kinkos. You can download and install software. When you are ready to print a document, you click on this, and off it goes. You can have it bound, hole punched, or enlarged.
So many uses for this. You can print a document to the Kinko's nearest a client and then the client can just pick it up. A virtual law clerk could research and write a memo and then have the attorney pick up a printed version. Law students could use this to print out papers and save the hassle of printing it at home, especially late at night the day before it is due when printers have a habit of breaking.
This is a great little program that you can use.
Many thanks, Jonathan and Terry!
Ban Xenical, Public Citizen urges
Public Citizen petitioned the U.S. Food and Drug Administration to remove from the market the prescription version of Xenical (orlistat, made by Roche Pharmaceuticals) because the obesity treatment has been known to cause a significant increase in aberrant crypt foci (ACF), which are widely believed to be a precursor to colon cancer.
Last week, the FDA sent an “approvable” letter concerning the over-the-counter (OTC) version of orlistat to GlaxoSmithKline, which has applied to market this version. Despite safety concerns, if GlaxoSmithKline meets the conditions set by the FDA – conditions that have not been made public – this would make the drug eligible for final approval for OTC sales.
For more information, go here.
Last week, the FDA sent an “approvable” letter concerning the over-the-counter (OTC) version of orlistat to GlaxoSmithKline, which has applied to market this version. Despite safety concerns, if GlaxoSmithKline meets the conditions set by the FDA – conditions that have not been made public – this would make the drug eligible for final approval for OTC sales.
For more information, go here.
Monday, April 10, 2006
HIPAA Preemption Analysis by State
Thanks to the folks at the HIPAA blog for the link:
HIPAA Preemption Analysis of State Privacy and Security Laws: You can find it
here, at the site National Association of Chain Drug Stores Foundation.
For example, for Georgia you can find this:
Are there any statutes or regulations governing the privacy of a customer’s personal information used or disclosed by a retail pharmacy?
Yes, in Georgia there are privacy-related statutory and regulatory requirements in the following areas:
· Confidential Information
· Disclosure
· Pharmacist-Patient Privilege
· HIV/AIDS
In addition, there are information-related statutory and regulatory requirements in the following areas:
· Record Maintenance
· Records of Dispensing
· Automated Data Processing Systems
· Transmission of Prescriptions
· Transfer of Prescriptions
HIPAA Preemption Analysis of State Privacy and Security Laws: You can find it
here, at the site National Association of Chain Drug Stores Foundation.
For example, for Georgia you can find this:
Are there any statutes or regulations governing the privacy of a customer’s personal information used or disclosed by a retail pharmacy?
Yes, in Georgia there are privacy-related statutory and regulatory requirements in the following areas:
· Confidential Information
· Disclosure
· Pharmacist-Patient Privilege
· HIV/AIDS
In addition, there are information-related statutory and regulatory requirements in the following areas:
· Record Maintenance
· Records of Dispensing
· Automated Data Processing Systems
· Transmission of Prescriptions
· Transfer of Prescriptions
Friday, April 07, 2006
Vioxx: Gilmartin on the stand
It's the first testimony in any trial setting by Merck's former chief, Raymond Gilmartin. He testified in the punitive damages phase of the McDarby trial.
You can read much more by going to the Forbes site, here.
From the Philly paper:
Gilmartin appeared to be taken off-guard once when attorney Mark Lanier showed him an e-mail from his onetime chief scientist that appeared to question Gilmartin's leadership. Edward Scolnick wrote an email to new CEO Peter Kim, expressing hope for a "reborn" Merck under a "new CEO" who would not restrict Kim's freedom as Scolnick's had been restricted "in recent years."
Gilmartin, was shown that email after he had finished testifying that Scolnick was a great scientist dedicated to safety. He paused when the email was discussed.
Go here for more.
From the Forbes article:
During the hearing, Lanier showed jurors an internal analysis Merck performed in October 2000. It pooled data from clinical studies and allegedly indicated that Vioxx users suffered more than twice as many heart attacks as those on comparison drugs used in the studies.
The attorney declared that the analysis was never given to the U.S. Food and Drug Administration. "So you are telling us it was important enough to do the study but not important enough to send to the FDA?" Lanier asked him.
"No, I'm not telling you that at all," the AP quoted Gilmartin as snapping back.
You can read much more by going to the Forbes site, here.
From the Philly paper:
Gilmartin appeared to be taken off-guard once when attorney Mark Lanier showed him an e-mail from his onetime chief scientist that appeared to question Gilmartin's leadership. Edward Scolnick wrote an email to new CEO Peter Kim, expressing hope for a "reborn" Merck under a "new CEO" who would not restrict Kim's freedom as Scolnick's had been restricted "in recent years."
Gilmartin, was shown that email after he had finished testifying that Scolnick was a great scientist dedicated to safety. He paused when the email was discussed.
Go here for more.
From the Forbes article:
During the hearing, Lanier showed jurors an internal analysis Merck performed in October 2000. It pooled data from clinical studies and allegedly indicated that Vioxx users suffered more than twice as many heart attacks as those on comparison drugs used in the studies.
The attorney declared that the analysis was never given to the U.S. Food and Drug Administration. "So you are telling us it was important enough to do the study but not important enough to send to the FDA?" Lanier asked him.
"No, I'm not telling you that at all," the AP quoted Gilmartin as snapping back.
Thursday, April 06, 2006
On the Cona Verdict - Attorneys' Fees Award
For those out there that think that Mark Lanier will only earn a contingency fee from the $135 dollar verdict ... he will seek substantially more.
The NJ statute dealing with consumer fraud allows for an application of attorneys' fees. Each case had a consumer fraud count which was ruled on in favor of the Plaintiff.
You can surely expect that in Lanier's application for fees, he may seek more than $400,000.
The NJ statute dealing with consumer fraud allows for an application of attorneys' fees. Each case had a consumer fraud count which was ruled on in favor of the Plaintiff.
You can surely expect that in Lanier's application for fees, he may seek more than $400,000.
Vioxx Verdicts: For Plaintiffs
Well, sort of. By now most are digesting the impact of the verdicts yesterday in New Jersey. The McDarby case verdict exceeded $4 million, while the Cona case essentially was a zero (a jury award of $45, trebled to $135).
As they say in the South, the verdicts makes the claims against Merck "clear as mud."
A few thoughts:
* Merck now knows that there are two firms that can beat the Company in trial.
* Will Mark Lanier's fiery Texas style litigation work in New Jersey? It didn't work on the Cona case. He has an office now in New York, so he is in for the long haul.
* Did the N.Y. Firm of Weitz & Luxenburg have an East Coast style that plays better?
* Causation: Lanier took the approach that the marketing of the drug was the key part of fault here. That worked in his Texas verdict, but here the question was one of causation, as it should be.
The punitive damages phase on the McDarby case starts this morning, and as a result the damages could top $20 million.
Mr. McDarby is quite ill from all reports. He had a heart attack on April 15, 2004, at home in Park Ridge, N.J. McDarby said he took Vioxx daily for four years for arthritis. He is now confined to a wheelchair. He spent 34 days in the hospital and 69 days in a rehabilitation center before coming home. He cannot get up out of a chair, he can't get out of bed on his own, he can't dress himself and hHe needs assistance going to the bathroom.
What is signficant is that Mr. McDarby had significant risk factors when placed on Vioxx - his age, diabetes, high blood pressure, and clogged arteries. In December of 2004 many attorneys would have possibly turned down his case because of the risk factors. I saw a claim involving risk factors as one where Vioxx was in essence throwing gasonline on a fire. You can read the definitive study on risk factors in the Framingham Study.
The News: Once again lazy reporting on many TV outlets. Most reasonable attorneys would not see a $45 verdict as a "W." On a news report here in Atlanta last evenign, the sixty second blurb about the verdict included a teaser about two jury verdicts against Merck. The report said that Merck lost two cases in a State Court in New Jersy, including one verdict for more than four million dollars.
The real verdict on this litigation? Too early to tell. Mike Galpern is up next.
More later.
As they say in the South, the verdicts makes the claims against Merck "clear as mud."
A few thoughts:
* Merck now knows that there are two firms that can beat the Company in trial.
* Will Mark Lanier's fiery Texas style litigation work in New Jersey? It didn't work on the Cona case. He has an office now in New York, so he is in for the long haul.
* Did the N.Y. Firm of Weitz & Luxenburg have an East Coast style that plays better?
* Causation: Lanier took the approach that the marketing of the drug was the key part of fault here. That worked in his Texas verdict, but here the question was one of causation, as it should be.
The punitive damages phase on the McDarby case starts this morning, and as a result the damages could top $20 million.
Mr. McDarby is quite ill from all reports. He had a heart attack on April 15, 2004, at home in Park Ridge, N.J. McDarby said he took Vioxx daily for four years for arthritis. He is now confined to a wheelchair. He spent 34 days in the hospital and 69 days in a rehabilitation center before coming home. He cannot get up out of a chair, he can't get out of bed on his own, he can't dress himself and hHe needs assistance going to the bathroom.
What is signficant is that Mr. McDarby had significant risk factors when placed on Vioxx - his age, diabetes, high blood pressure, and clogged arteries. In December of 2004 many attorneys would have possibly turned down his case because of the risk factors. I saw a claim involving risk factors as one where Vioxx was in essence throwing gasonline on a fire. You can read the definitive study on risk factors in the Framingham Study.
The News: Once again lazy reporting on many TV outlets. Most reasonable attorneys would not see a $45 verdict as a "W." On a news report here in Atlanta last evenign, the sixty second blurb about the verdict included a teaser about two jury verdicts against Merck. The report said that Merck lost two cases in a State Court in New Jersy, including one verdict for more than four million dollars.
The real verdict on this litigation? Too early to tell. Mike Galpern is up next.
More later.
Tuesday, April 04, 2006
Vioxx Trial: Desperate Executives?
Plaintiff's Attorney Mark Lanier captivated the courtroom yesterday in closing argument on the Cona case.
Lanier presented his 70-minute closing statement to the jury in the form of a TV series he called 'Desperate Executives' - a takeon the popular series "Desperate Housewives." Lanier's fictional series consisted of four episodes: "Shoot for the Moon"; "Trouble in Paradise"; "The Cover Up"; and "Game Over."
The story, Lanier said, began with the desire to make money from Vioxx, progressed through a series of evasions and denials that the drug caused heart attacks, and ended with Vioxx being criticized by a number of scientific reports.
Great stuff. Read more here.
Lanier presented his 70-minute closing statement to the jury in the form of a TV series he called 'Desperate Executives' - a takeon the popular series "Desperate Housewives." Lanier's fictional series consisted of four episodes: "Shoot for the Moon"; "Trouble in Paradise"; "The Cover Up"; and "Game Over."
The story, Lanier said, began with the desire to make money from Vioxx, progressed through a series of evasions and denials that the drug caused heart attacks, and ended with Vioxx being criticized by a number of scientific reports.
Great stuff. Read more here.
Monday, April 03, 2006
Vioxx: Cona closing arguments
Closing arguments begin today:
A Merck lawyer told the jury in the Cona joint trial that there was no evidence in the latest Vioxx trial that the painkiller caused the heart attacks of two men who have sued the drug maker.
Christy Jones said the two men had coronary artery disease, and one of them had diabetes, a risk factor for heart disease. Jones also said Merck acted responsibly in studying Vioxx's risks and warned of the risks in the drug's label.
Jones walked jurors through a timeline of studies of Vioxx since it was launched in the U.S. in 1999. Merck followed up on studies suggesting Vioxx had risks, and submitted all data to the FDA. The FDA itself approved additional uses for Vioxx even with these risks, which were added to the drug's prescription label, Jones said. Plaintiffs' attorneys are set to make closing statements later Monday, after which the case will go to the jury.
A Merck lawyer told the jury in the Cona joint trial that there was no evidence in the latest Vioxx trial that the painkiller caused the heart attacks of two men who have sued the drug maker.
Christy Jones said the two men had coronary artery disease, and one of them had diabetes, a risk factor for heart disease. Jones also said Merck acted responsibly in studying Vioxx's risks and warned of the risks in the drug's label.
Jones walked jurors through a timeline of studies of Vioxx since it was launched in the U.S. in 1999. Merck followed up on studies suggesting Vioxx had risks, and submitted all data to the FDA. The FDA itself approved additional uses for Vioxx even with these risks, which were added to the drug's prescription label, Jones said. Plaintiffs' attorneys are set to make closing statements later Monday, after which the case will go to the jury.
Subscribe to:
Posts (Atom)